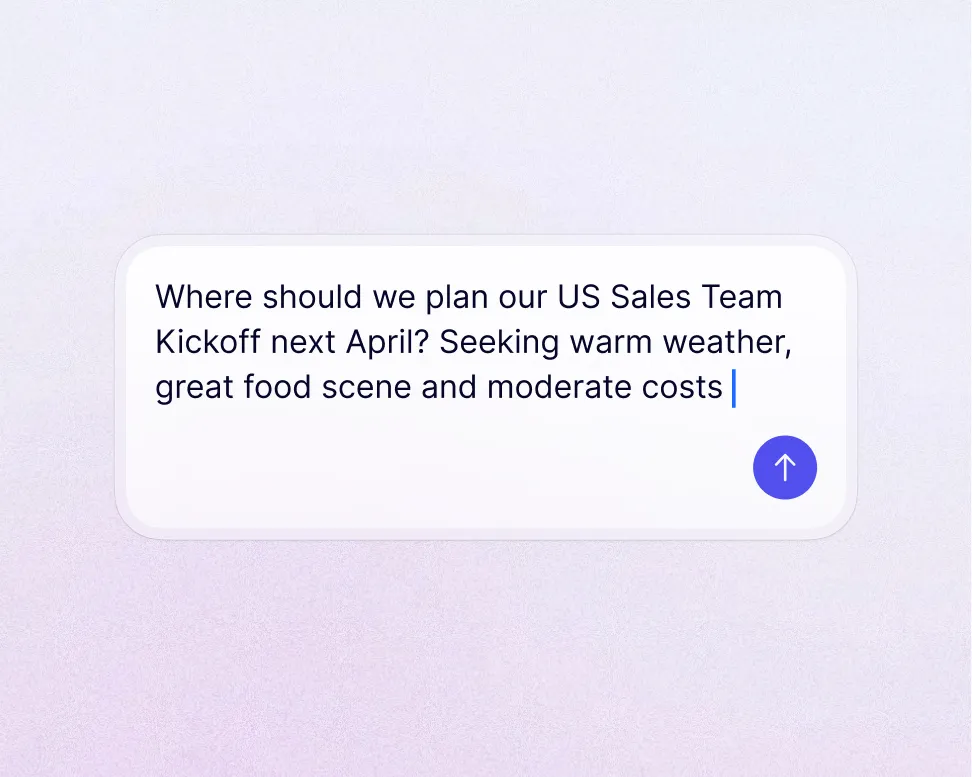
Travel program management tailored to pharma companies
Clarasight normalizes travel, expense, and card data across therapeutic areas, business units, and geographies into one canonical model, so your team maintains compliance, controls spend, and reports on emissions with confidence.

Trusted by modern enterprise teams running complex global travel programs






























Eyebrow
Your clinical and commercial travel is at a scale that spreadsheets can't keep up with.
In pharma, congresses, launches, HCP travel, and field activity create demand spikes most programs still manage reactively.

Congresses and events require travel
Congresses and major events are often planned elsewhere but still drive booking and cost.

The compliance burden is immense
When HCP and non-employee data is fragmented, reporting becomes a legal risk.

Travel demand spikes hit fast
Launches, trials, and commercial activity create travel volume that arrives fast.

Eyebrow
Travel at Pharma Companies is part of commercial delivery, not just program overhead.
In pharmaceuticals, travel is unusually hard to separate from how the business operates. Field teams travel to support commercial activity. Congresses and conferences are critical for presenting data, meeting with KOLs and payers, and advancing relationships. Internal meetings still happen on top of that. Add non-employee travel and HCP compliance requirements, and travel becomes far more complex than a standard corporate program.


Simulate congress and launch demand
Unify event travel planning
Bring congress, internal meeting, and BAU travel into one planning view.
Forecast and predict travel spikes
Anticipate trial milestones and launch periods before demand hits unexpectedly.
Manage budgets with rigor
Forecast, run scenarios and plan across teams with agility





Capture HCP spend at booking
Plan and manage
Record compliance data during booking instead of rebuilding it from expense reports later.
Track TOV
Keep HCP spend and reporting fields tied to each trip and meeting.
Drastically reduce manual work
Reduce manual effort to reconcile data by 90% and reporting time by 99%

Eyebrow
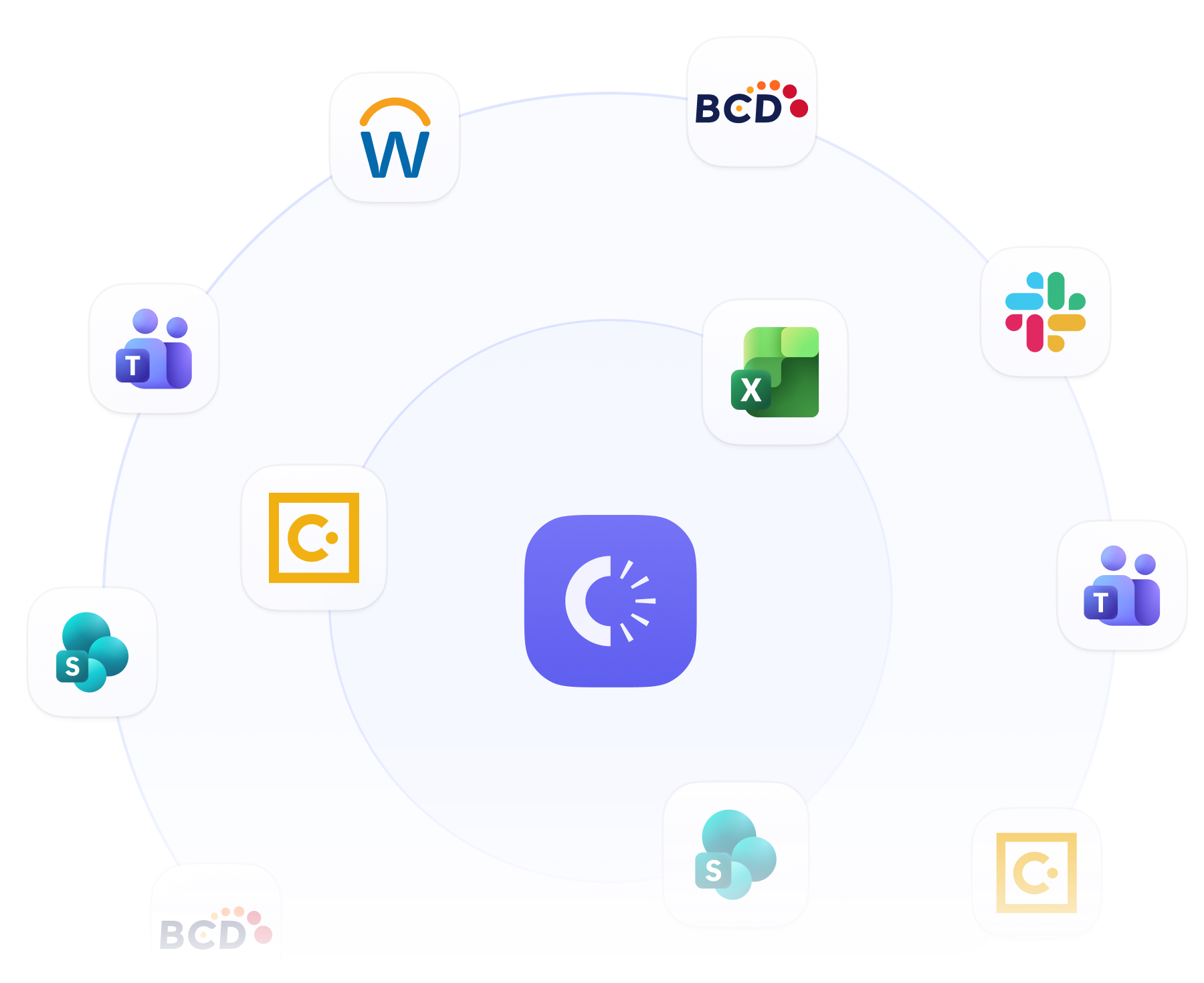
Your stack, connected. Your data, unified.
Clarasight sits between your existing systems, automating the data mapping and workflows that connect them.
Connects to your TMC, expense, card, HR, and OBT systems
One canonical model across every source
No rip-and-replace required

Your data is mission critical.
We protect it that way.
Role-based access controls and permissions
End-to-end encryption in transit and at rest
Full audit logging and activity traceability

ISO 27001

ISO 27701

SOC 2

GDPR

Dedicated expert teams who build and deploy with you.
Eyebrow
Questions? Answers.
Your most frequently asked questions, all in one place. If you don’t see what you need, reach out to us.
How does Clarasight help pharmaceutical companies manage the complexity of global clinical and commercial travel programs?
Clarasight unifies travel, expense, card, and HR data into one canonical model, giving pharmaceutical companies consolidated visibility across clinical, commercial, and corporate travel activity without manual reconciliation across mismatched systems.
Can pharmaceutical companies use Clarasight to enforce travel policy and expense controls across highly regulated environments?
Yes. Clarasight monitors spend against policy in real time, automatically routing exceptions for approval and logging every action, supporting the governance and auditability requirements pharmaceutical companies face across regulatory and compliance frameworks.
How does Clarasight support pharmaceutical companies in tracking and reporting on travel emissions for sustainability commitments?
Clarasight calculates travel emissions across air, ground, and hotel spend using recognized methodologies, giving pharmaceutical companies the credible, audit-ready data needed for ESG reporting, net zero target tracking, and regulatory disclosure.
Does Clarasight integrate with the systems pharmaceutical companies already use to manage travel and expense?
Yes. Clarasight connects to your existing TMC, OBT, expense platform, card provider, and HR system, normalizing data from every source into one unified model. No rip-and-replace required and no disruption to existing infrastructure.
How does Clarasight help pharmaceutical companies manage travel spend across multiple business units, therapeutic areas, and geographies?
Clarasight normalizes data across business units, cost centers, and geographies into one model, giving pharmaceutical companies consolidated visibility and consistent policy enforcement across complex, globally distributed programs.
How does Clarasight support pharmaceutical companies in demonstrating travel program value to finance and executive leadership?
Clarasight surfaces measurable program outcomes including cost savings, policy compliance rates, and vendor performance metrics, giving travel and procurement leaders the evidence needed to demonstrate program ROI to finance and executive stakeholders.